noun
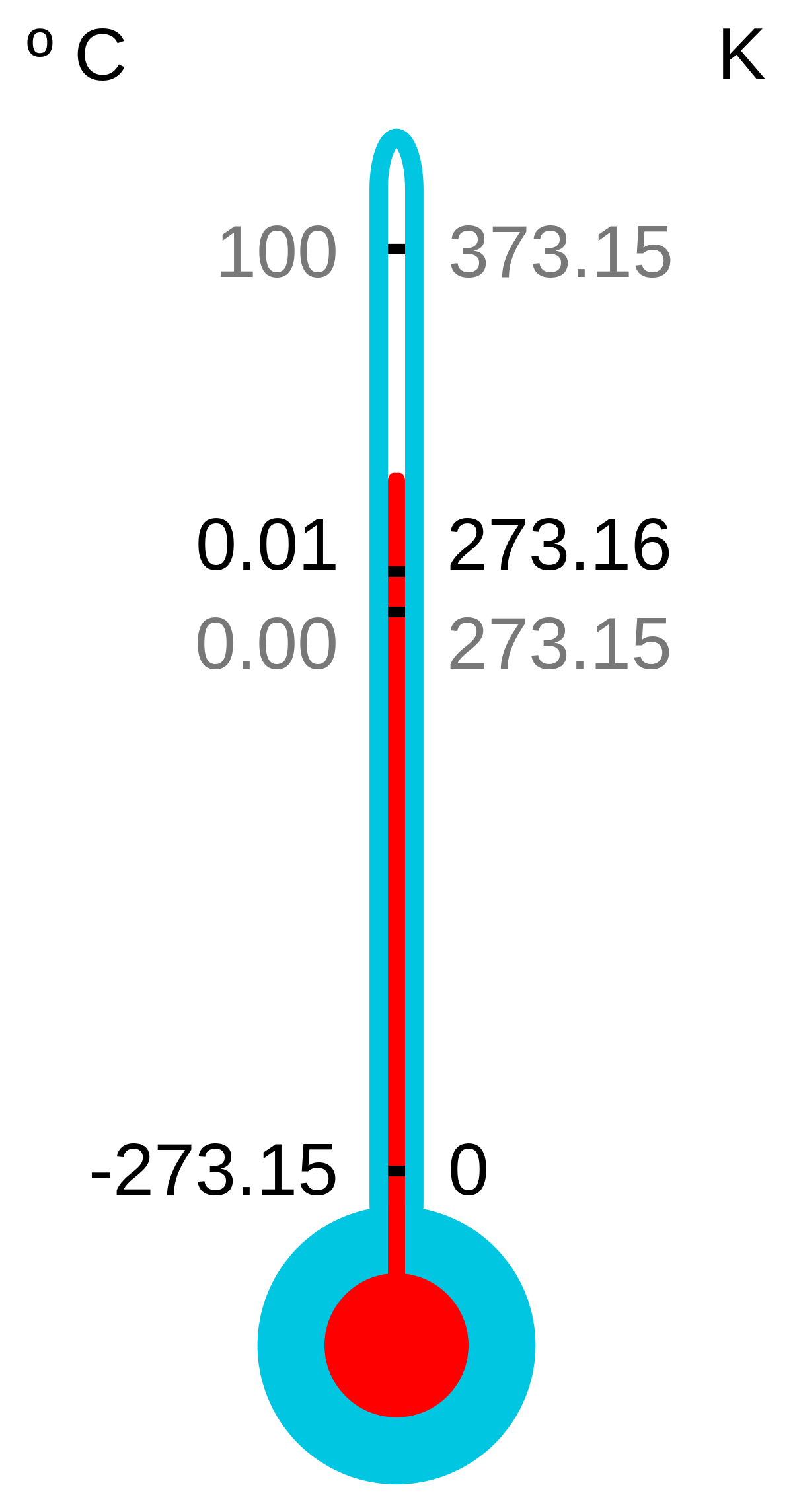
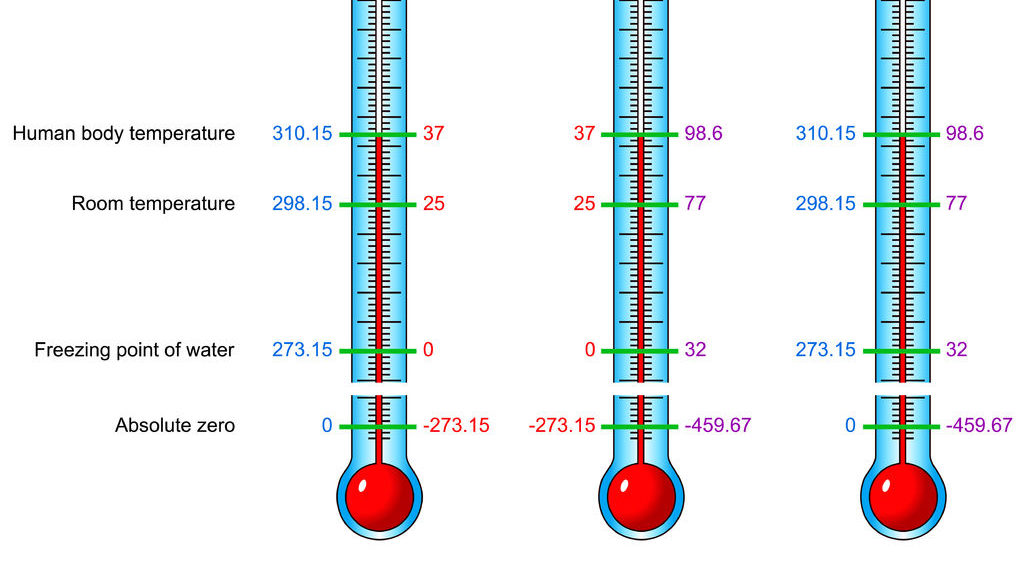
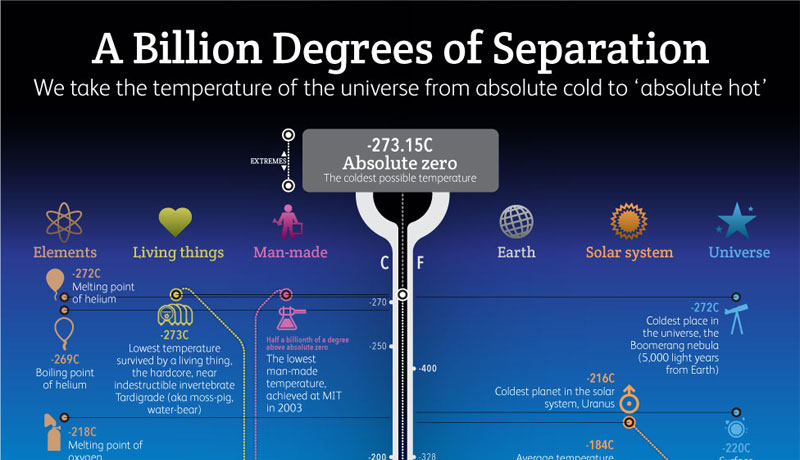
- the temperature of −273.16°C (−459.69°F), the hypothetical point at which all molecular activity ceases.
noun Thermodynamics.
- temperature (absolute temperature) as measured on a scale in which the hypothetical lowest limit of physical temperatures is assigned the value zero (absolute zero), as the Kelvin scale.
noun
- the lowest temperature theoretically attainable, at which the particles constituting matter would be in the lowest energy states available; the zero of thermodynamic temperature; zero on the International Practical Scale of Temperature: equivalent to –273.15°C or –459.67°F
the idea dates back to 1702 and its general value was guessed to within a few degrees soon thereafter, but not precisely discovered until Lord Kelvin’s work in 1848. It was known by many names, e.g. infinite cold, absolute cold, natural zero of temperature; the term absolute zero was among them by 1806.
n.
- The temperature at which substances possess no thermal energy, equal to -273.15°C, -459.67°F, or 0 K.
- A temperature scale having absolute zero as the lowest temperature. Absolute temperature scales only have positive numbers. The Kelvin scale and the Rankine scale are absolute temperature scales. Compare relative temperature scale.
- The lowest possible temperature, at which all molecules are have the least possible amount of kinetic energy. Absolute zero is equal to 0°K, -459.67°F, or -273.15°C. At temperatures approaching absolute zero, the physical characteristics of some substances change significantly. For example, some substances change from electrical insulators to conductors, while others change from conductors to insulators. Absolute zero has never been reached in laboratory experiments. See also Bose-Einstein condensate zero-point energy.
The lowest temperature that can be attained by matter, corresponding to the point at which most motion in atoms stops. Absolute zero is about –273 degrees on the Celsius scale and about –460 on the Fahrenheit (see also Fahrenheit) scale.
 Liberal Dictionary English Dictionary
Liberal Dictionary English Dictionary