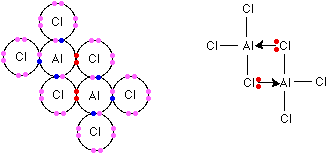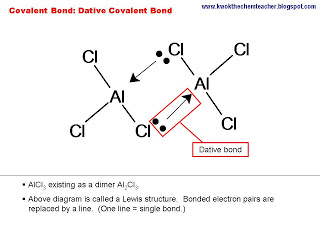
noun Chemistry.
- a type of covalent bond between two atoms in which the bonding electrons are supplied by one of the two atoms.
noun
- chem another name for coordinate bond
noun
- a type of covalent chemical bond in which both the shared electrons are provided by one of the atomsAlso called: dative bond, semipolar bond
n.
- A covalent chemical bond between two atoms that is produced when one atom shares a pair of electrons with another atom lacking such a pair.semipolar bond
- See coordinate bond.
- A type of covalent bond in which both the shared electrons are contributed by one of the two atoms. Also called dative bond See more at covalent bond.
 Liberal Dictionary English Dictionary
Liberal Dictionary English Dictionary