eutectic [yoo-tek-tik]Physical Chemistry ExamplesWord Origin adjective
- of greatest fusibility: said of an alloy or mixture whose melting point is lower than that of any other alloy or mixture of the same ingredients.
- noting or pertaining to such a mixture or its properties: a eutectic melting point.
noun
- a eutectic substance.
Origin of eutectic 1880–85; Greek eútēkt(os) easily melted, dissolved (eu- eu- + tēktós melted) + -ic Examples from the Web for eutectic Historical Examples of eutectic
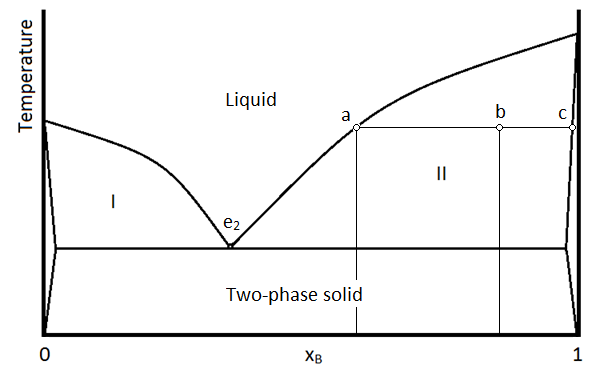
From our curve we should expect a eutectic mixture with about 35 per cent.
Scientific American Supplement, No. 508, September 26, 1885
Various
When the eutectic point is reached, the temperature remains constant, until all the liquid has solidified.
The Phase Rule and Its Applications
Alexander Findlay
The length of time during which the temperature remains constant at the point c, depends, of course, on the eutectic solution.
The Phase Rule and Its Applications
Alexander Findlay
If the initial composition of the magma is represented by a point between the two eutectic points, orthoclase will separate first.
The Phase Rule and Its Applications
Alexander Findlay
After this the temperature again falls, until it again remains constant at the eutectic point c.
The Phase Rule and Its Applications
Alexander Findlay
British Dictionary definitions for eutectic eutectic adjective
- (of a mixture of substances, esp an alloy) having the lowest freezing point of all possible mixtures of the substances
- concerned with or suitable for the formation of eutectic mixtures
noun
- a eutectic mixture
- the temperature on a phase diagram at which a eutectic mixture forms
Word Origin for eutectic C19: from Greek eutēktos melting readily, from eu- + tēkein to melt eutectic in Science eutectic [yōō-tĕk′tĭk]
- The proportion of constituents in an alloy or other mixture that yields the lowest possible complete melting point. In all other proportions, the mixture will not have a uniform melting point; some of the mixture will remain solid and some liquid. At the eutectic, the solidus and liquidus temperatures are the same.
- An alloy or other mixture with constituents in the proportions of the eutectic.
- The melting point of the eutectic.
 Liberal Dictionary English Dictionary
Liberal Dictionary English Dictionary