noun Chemistry.
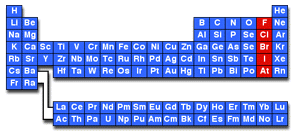
- any of the electronegative elements, fluorine, chlorine, iodine, bromine, and astatine, that form binary salts by direct union with metals.
noun
- any of the chemical elements fluorine, chlorine, bromine, iodine, and astatine. They are all monovalent and readily form negative ions
general name for elements of the chlorine family, 1842, from Swedish, coined by Swedish chemist Baron Jöns Jakob Berzelius (1779-1848), literally “salt-producer,” from Greek hals “salt” (see halo-) + -gen “giving birth to” (see -gen); so called because a salt is formed in reactions involving these four elements.
n.
- Any of a group of five chemically related nonmetallic elements including fluorine, chlorine, bromine, iodine, and astatine.
- Any of a group of five nonmetallic elements with similar properties. The halogens are fluorine, chlorine, bromine, iodine, and astatine. Because they are missing an electron from their outermost shell, they react readily with most metals to form salts. See Periodic Table.
 Liberal Dictionary English Dictionary
Liberal Dictionary English Dictionary



