noun
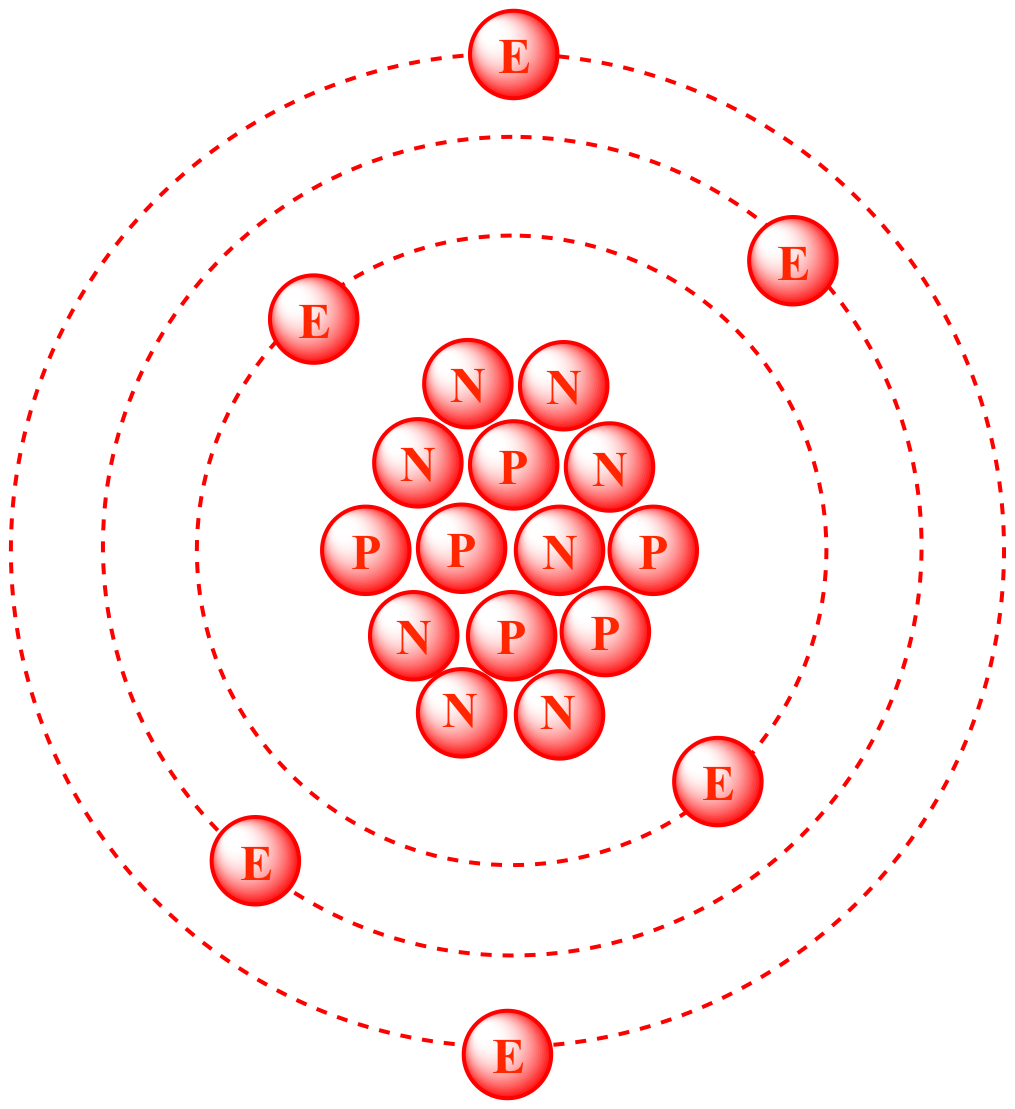
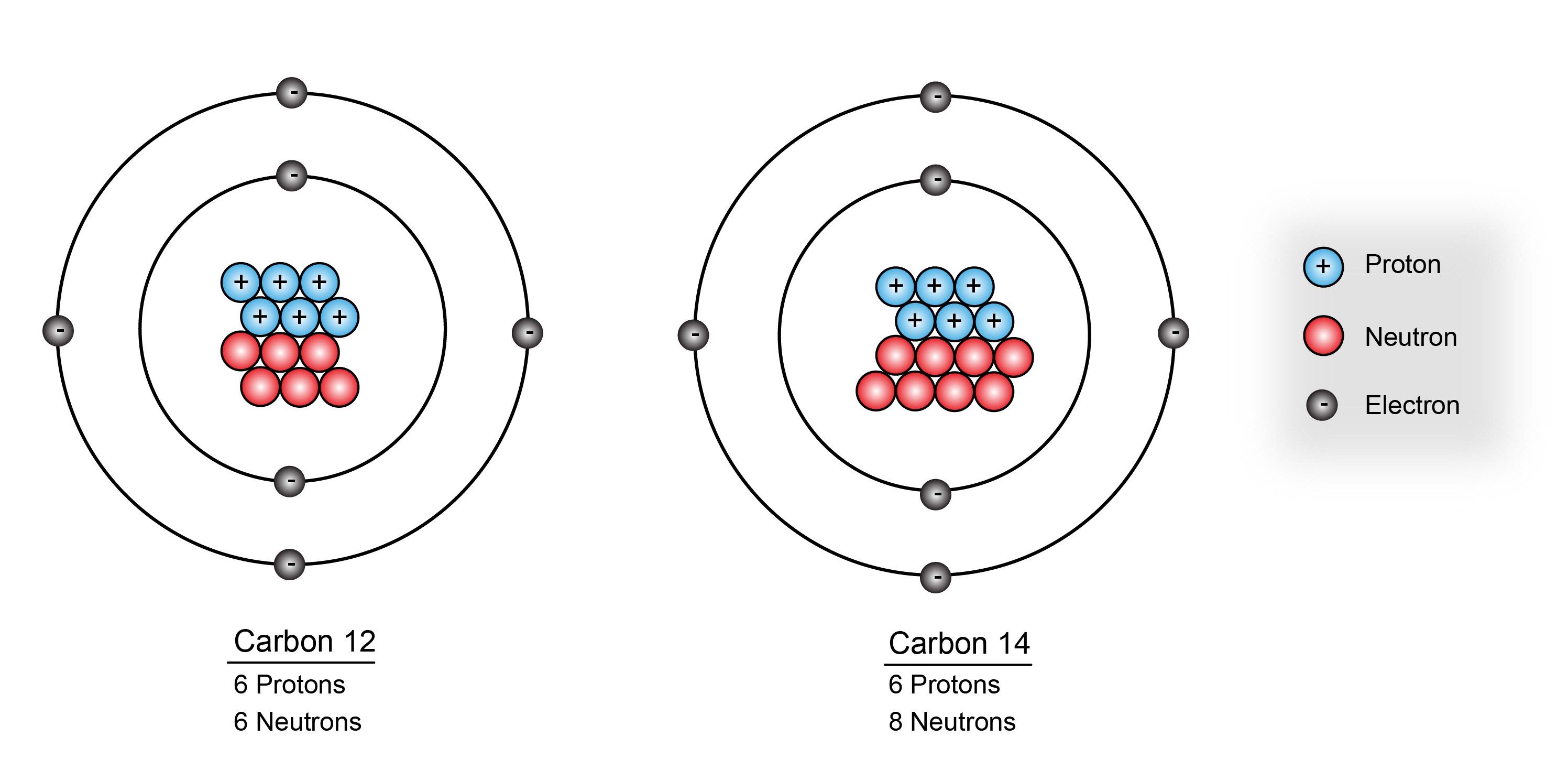
- A naturally occurring radioactive isotope of carbon having six protons and eight neutrons in the nucleus. Carbon 14 is important in dating archaeological and biological remains by radiocarbon dating.
A radioactive isotope of carbon. A carbon 14 atom contains six protons, six electrons, and eight neutrons. Carbon 14 is produced when neutrons bombard atoms of nitrogen.
 Liberal Dictionary English Dictionary
Liberal Dictionary English Dictionary